 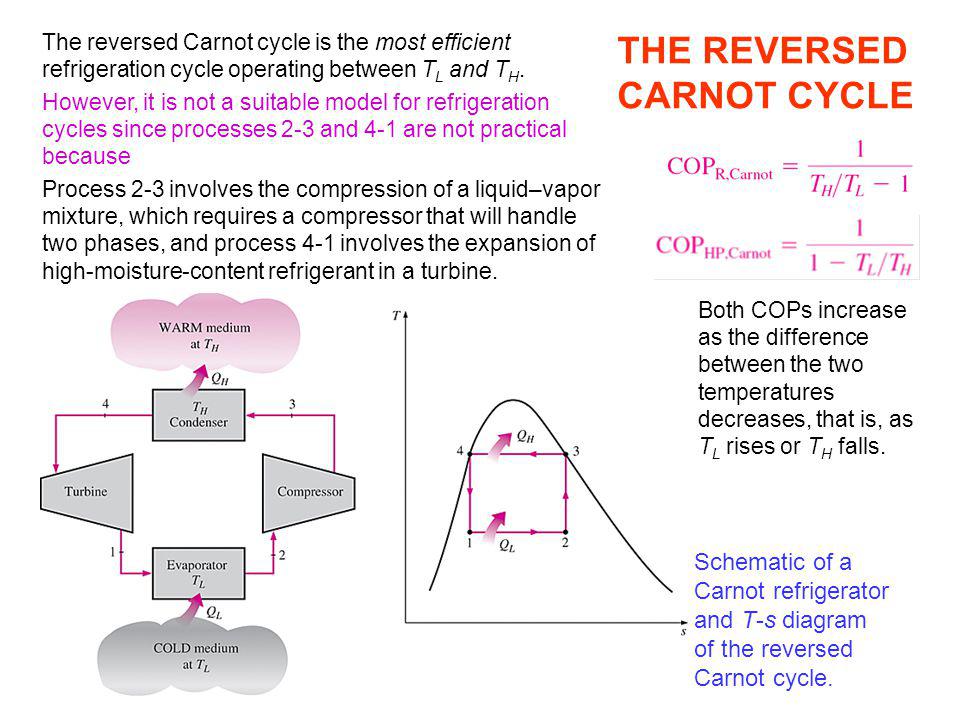
The second law provides the necessary means to determine the quality as well as the degree of degradation of energy during a process. The first law is concerned with the quantity of energy and the transformations of energy from one form to another with no regard to its quality. 
The second law also asserts that energy has quality. The second law may be used to identify the direction of processes. A process must satisfy both the first and second laws of thermodynamics to proceed. Processes occur in a certain direction, and not in the reverse direction. These processes cannot occur even though they are not in violation of the first law. Transferring heat to a paddle wheel will not cause it to rotate. 
Transferring heat to a wire will not generate electricity. Introduction to the Second Law Introduction to the Second Law A cup of hot coffee does not get hotter in a cooler room. Determine the expressions for the thermal efficiencies and coefficients of performance for reversible heat engines, heat pumps, and refrigerators. Examine the Carnot principles, idealized Carnot heat engines, refrigerators, and heat pumps. Apply the second law to develop the absolute thermodynamic temperature scale. 
Apply the second law of thermodynamics to cycles and cyclic devices. Discuss the concepts of perpetual-motion machines. Describe the Kelvin Planck and Clausius statements of the second law of thermodynamics. Discuss thermal energy reservoirs, reversible and irreversible processes, heat engines, refrigerators, and heat pumps. Identify valid processes as those that satisfy both the first and second laws of thermodynamics. A., Thermodynamics: An Engineering Approach, McGraw-Hill, New York, 6th Ed., 2008 Introduce the second law of thermodynamics. 1 Objectives MAE Chapter 6 The Second Law of Thermodynamics The content and the pictures are from the text book: Çengel, Y. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
